Doliolids are tiny marine animals so transparent they are practically invisible. Yet, these rarely seen and little understood organisms are a major driver of the marine ecosystem on continental shelves around the world. University of Georgia researcher Marc Frischer recently completed a major field study in the South Atlantic Bight on the continental shelf off the coast of Georgia in an effort to understand these mysterious animals. The study, funded by the U.S. National Science Foundation, involved 25 research cruises over a two-and-a-half-year period.
Doliolids are gelatinous zooplankton that, as adults, are about the size of a pencil eraser. They live in the subsurface water column on continental shelves. Although they are almost always present on the shelf, their populations fluctuate dramatically. Sometimes they are difficult to find while at other times, when the conditions are right, there can be thousands of them in a cubic meter (264 gallons) of water.
“You can almost walk across them,” Frischer said.

A microscopic view of a doliolid.
When this happens, they consume nearly all the production of microscopic marine plants known as phytoplankton on the continental shelf. Continental shelves account for about eight percent of the ocean but generate half of all ocean biological productivity.
“When doliolids are blooming it’s a completely different regime in terms of the food web,” Frischer said.
Although, at times, doliolids may be a dominant force in the marine food web, they remain mysterious and extremely understudied.
“We don’t even have a good idea of what all eat,” Frischer said. “So we cannot make predictions of what their impacts are going to be, what changes we might expect to see and what that means. All those questions are still unknown, which is why we are trying to answer them.”

Marc Frischer (left) prepares a water collection device along with R/V Savannah marine technician Zach Tait.
Being small and nearly transparent, doliolids are difficult to study. They rarely, if ever, come to the surface, making it very difficult to observe them in their natural setting. Because they are gelatinous, like a piece of jelly, it is also difficult to bring them back to the lab to study. Even if brought back to the lab successfully, researchers cannot replicate all of the intricacies of the ocean environment. So, to understand the doliolids’ diet, Frischer and his team turned to the tools of molecular biology. They analyze DNA.
“We capture a doliolid that has been feeding and remove all of the doliolid’s DNA from the sample,” Frischer said. “Everything that is left is either what it has been eating or a parasite. We can identify and rule out the parasite DNA and what remains is what it ate.”
Over the course of 30 months, Frischer and his team have learned a lot about doliolids in their natural environment.
“We were surprised that doliolids were nearly always present, how much their population size can vary, and that their blooms are relatively frequent,” Frischer said.
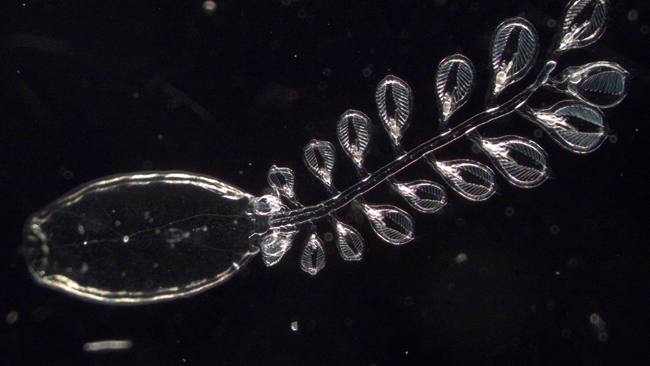
A doliolid with a tail of smaller doliolids which are feeding the larger organism.
These blooms occur primarily during the summer and winter when seasonal weather patterns stir up the water and pull nutrient-rich cold water up from the ocean depths and fertilize the sunlit surface waters. This fertilization process allows phytoplankton, one of doliolids’ primary foods, to grow prolifically.
Another surprise was that doliolids can be picky eaters. Researchers have always thought doliolids were passive feeders, but Frischer and his team have found they may also have the ability to select what they eat. Doliolids capture their prey by filtering water. They suck in water and they catch what’s in it. When analyzing the doliolids’ gut contents, the researchers discovered much higher concentrations of some types of prey inside doliolids relative to the concentration of that prey type in the surrounding water.
“For doliolids, one of the more nutritious types of phytoplankton are diatoms,” Frischer said. “We found they can capture a million-fold more diatoms, and capture them preferentially, than what’s in the general water column. We don’t know yet how they do that.”
Frischer speculates that because doliolids, particularly when they are present in high abundances, filter massive volumes of water and can seemingly specifically capture certain prey types that they may have an extremely large influence on the marine food web. Frischer said that doliolids appear to be capable of structuring the pathway of energy and matter in productive continental shelf systems. For example, Frischer’s team observed that copepod DNA was nearly always found inside doliolids. Copepods are small shrimp-like animals and they account for the largest portion of the animal biomass in the oceans.
“We doubt that doliolids can capture adult copepods, but they could easily capture the eggs and larvae,” Frischer said.
Frischer suspects that a major doliolid bloom could remove enough copepod eggs to effect the size of copepod populations allowing doliolids to outcompete copepods for the plankton prey that they both consume.
“Copepods are the primary food source for many marine animals, including small fish, so that the dynamic between copepods and doliolids is certainly an interesting one,” he said.
The researchers are now analyzing their data and preparing to publish their results in the peer-reviewed literature. Frischer says that as with most good research projects, their work on doliolids has raised many more questions than it has answered. “Doing these kinds of studies, with slow and careful effort that can be replicated, enables us to address and continue to understand more and more about how our ocean systems work.”