This cruise adventure began with pouring rain as we scrambled to wire everything for our plankton imaging system (mini Deep Focus Plankton imager, mDPI). Our mDPI had experienced technical issues during the previous two attempts to deploy it at-sea, but we were (cautiously) optimistic this time with the improved connection durability from a ‘new’ UNOLS fiber-optic winch. Into the afternoon of day 1, the weather improved, although the R/V Savannah would regularly surprise us with a deep roll over a seemingly non-existent swell on the glassy-surfaced sea. Looking back on previous years’ cruise reports, we once again found doliolids in February but not January; which makes it interesting that they have appeared in one or the other, but not both.
As the goal of this cruise was to capture and culture Dolioletta gegenbauri, the majority of our attention was focused on sampling at the 45 m isobath. According to Marc’s experience, doliolids can sometimes be found at the more oceanic station (45 m) and not at the more in-shelf one (25 m), so besides field-testing our equipment and examining the contents of a net-tow, we only briefly spent time at the 25 m station this cruise. On-ship analysis of the images from the mDPI revealed that we were still having some issues with the lens/light-camera alignment inside the imager, as there was an apparent shadow. We did see some doliolids, although not in any large numerical abundance. Hydromedusae of several varieties were observed in numbers, specifically Solmaris sp. (not pictured here, but see image at bottom for an example hydromedusa).
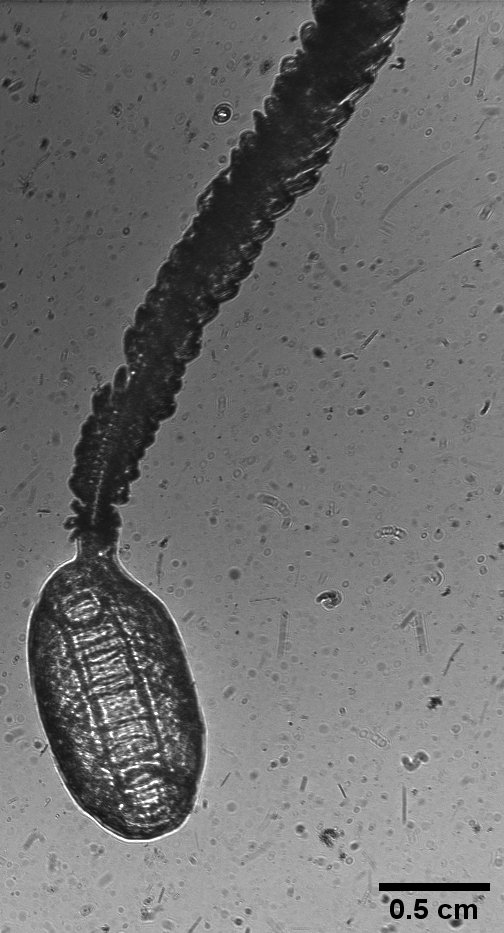
Doliolid nurse (Dolioletta gegenbauri with large cadophore “tail”). The buds on the tail feed to nourish the entire colony.
At the more oceanic station, both our net tows and imager deployments yielded successful results. We collected and isolated several (~7) nurse Dolioletta gegenbauri (pictured above) along with numerous other life stages. Although some died shortly after isolation, many survived to this day and are reproducing (as of February 20) in cultures maintained by Marc (with continued assistance from Tina). We’ll use this culture for experimental lab studies in the near future; stay tuned. In addition, from the tow, an eel larva (Leptocephalus) was collected and imaged on the benchtop imager before release.
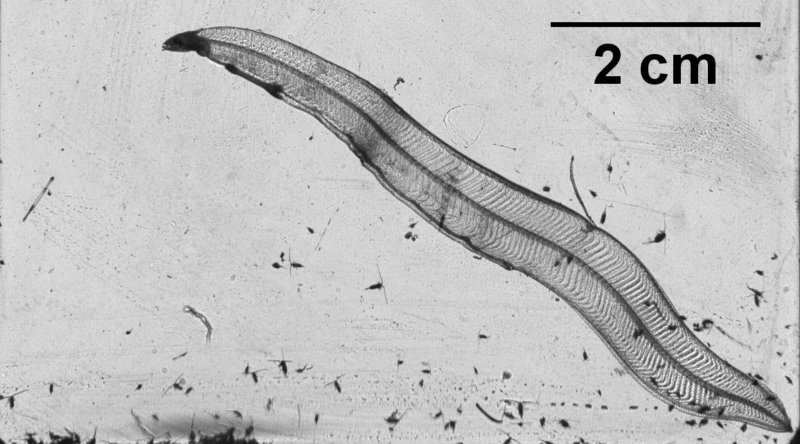
Leptocephalus larva (early stage eel) captured in the plankton net and imaged onboard with the benchtop camera system (same optics as the mDPI).
Post-cruise analyses of the images reveal that the mDPI does an excellent job at capturing some physical features that will be useful to monitor the life stage of doliolids as their internal organs disappear when they develop into nurses (pictured above). According to Marc, the abundance of doliolids we observed would fall into the ‘moderate bloom’ category. The images from the mDPI suggest that this ‘moderate bloom’ is likely to increase in number in the future – too bad we can’t go out next week to see if he was right. Numerous large nurses were observed, measuring upwards of 3-4 cm long with hundreds of trophozooids (the buds that feed to nourish the whole colony). I am excited at the potential information yielded by the combination of these image-datasets, physical data, microbial data, and isotope tracer data to reveal the workings and role of these bizarre creatures.

The Dol-MICROBE science crew poses by the mDPI imaging system on the RV Savannah (from left to right: Laura Treible, Patrick Duffy, Marc Frischer, and Adam Greer).
Future work in the Greer lab before the next cruise will entail re-examining the camera-light alignment issue, which only occurs when the sled is submerged, along with streamlining the processing and merging of physical and image data. Minor adjustments to cable position on the sled should solved problems with cables entering the camera field of view. It will be interesting to see how our work flow is when we also have to do numerous field-measurements that we need to achieve our long-term project goal of understanding the ecological function of doliolids on subtropical continental shelves and their influence on the microbial processes that shape our planet. If you are interested in reading more about the project please visit our project website and view the public abstract.
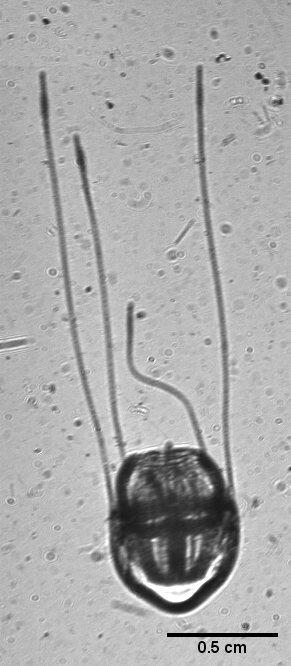
Large hydromedusa with 4 visible tentacles imaged with the mDPI.


One comment on “Cruise Log – R/V Savannah February 9-10, 2021 –DolEAGER2/DolMICROBE4: “Third Try’s the Charm” – by Patrick Duffy”