Doliolideers and friends,
Our first full DolMICROBE cruise is in the books! After months of waiting, training, and equipment testing, we finally received the green light to mount a full-scale research expedition on the R/V Savannah on April 5 & 6, 2021.The problem has been that, because of COVID-related safety precautions, we have not been able to include enough people to manage the full suite of measurements needed for the project. However, with COVID rates diminishing, and a greater confidence that the rigorous testing and distancing protocols we follow will keep the virus off the ship, we were finally able to give it a shot. Technically, although this was our 5th cruise associated with the project, it was actually our first full DolMICROBE cruise.

Full DolMICROBE science crew RV SAV 21-05 (DolMICROBE_05). Front row from the left: Patrick Duffy, Savannah Geiger, Cristin Archer. Back row from the left: Adam Greer, Laura Treible, Kyle Aaron, Anita Minniefield, Marc Frischer.
As Laura and Patrick described in our previous posts, the overarching goal of the DolMICROBE project is to test a hypothesis comparing periods when the food web is dominated by doliolids rather than crustacean zooplankton (e.g. copepods.) That is, when crustacean zooplankton are the primary consumers of phytoplankton, they tend to nourish larger specials (fish, birds, marine mammals), but when doliolids dominate the zooplankton communities, microbial loop processes are enhanced, resulting in a decrease in efficiency of trophic transfer to consumers.
In other words, when small gelatinous animals such as doliolids are abundant, energy and matter that would normally end-up producing fish and other large predators end-up shunted to microbes including bacteria and microscopic animals. To illustrate this hypothesis, take a look at this figure from our most recent paper published in the journal Limnology and Oceanography.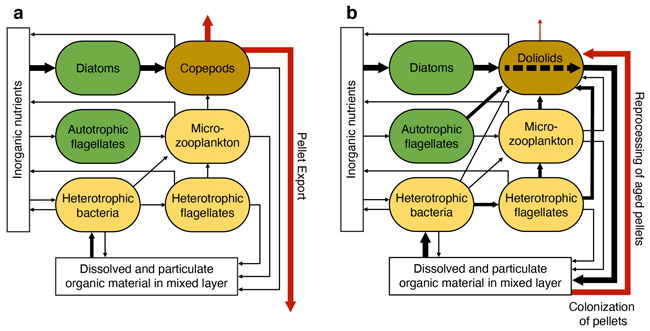 Hypothesized structure of pelagic food web when (A) copepods or (B) doliolids dominate zooplankton communities. Doliolid dominance is hypothesized to lead to increased complexity of the microbial network, decreased trophic transfer to higher trophic levels, and increased recycling of organic matter in shelf waters. The width of vector arrows indicates the hypothesized relative magnitude of flux. Red vector arrows indicate export flux pathways.
Hypothesized structure of pelagic food web when (A) copepods or (B) doliolids dominate zooplankton communities. Doliolid dominance is hypothesized to lead to increased complexity of the microbial network, decreased trophic transfer to higher trophic levels, and increased recycling of organic matter in shelf waters. The width of vector arrows indicates the hypothesized relative magnitude of flux. Red vector arrows indicate export flux pathways.
If you are interested in reading more you can download the full article HERE.
To test this hypothesis, our plan is to make measurements of many of the parameters represented by the ovals and arrows in the figure above. Our strategy is to repeatedly sample the same stations on the middle continental shelf of the South Atlantic Bight frequently enough to measure all of these parameters under varied conditions, and determine how the system functions differently when either copepods or doliolids dominate the zooplankton communities.
As is typical with all field oceanographic research, we are at the mercy of weather (COVID is a new challenge.), and this trip was no different. The cruise was originally scheduled for April 1 & 2 but the forecast was for 7 to 9-foot seas and 20 – 25 knots of wind from the Northwest -basically a Nor’Easter. When it is that rough, we’d have a very hard time getting the work done, and we’d probably all be seasick. We decided to wait for better weather. Delaying the trip turned out to be a blessing, not only because of the weather, but it gave us an opportunity to repair some equipment that we hadn’t realized was broken. Getting ready for the cruise, Adam discovered that the fiber optic connection to the in situ plankton imager had been damaged, and he and his team had to re-do the connection, with critical help from our resident IT person, Wayne Aaron, the R/V Savannah’s marine technician, Morgan Hudgins and our marine operations lead, John Bichy.. Adam had to order some parts and materials, so it was good that we had a few extra days to fix this issue. It also took the microbiology team longer than usual to load the ship. A lot of what we are doing is new, so it took us a while to pull it all together despite all the preparation that we had done in the lab.
By the time we were finally able to go, everything was fixed and organized. We were also rewarded with extremely pleasant weather and seas. The seas ended up being only 2 to 3’ with a gentle breeze from the SW (warm tropical air rather than cold Artic air).
Leaving the dock at 8:00 am as scheduled, we had a hearty breakfast prepared by chef Ben Leland who, in my opinion, is one of the best chefs in the fleet.

Chef Ben Leland serves Cristin Archer and Savannah Geiger breakfast.
Following breakfast, first mate Cam Albrant provided a safety briefing, and the ship conducted a man overboard drill. One thing that I am always impressed with on the R/V Savannah is their dedication to safety and their common-sense approach to making sure that we are all safe while at sea. Because the ship is so well taken care of and the crew well trained, safety is one thing that I don’t have to worry about.
Because we wanted to avoid too much congregation on the ship, we had held our science briefing prior to the cruise on Zoom.
For the rest of the morning, while we steamed out of the estuary and across the inner part of the continental shelf. We used the time to familiarize the science crew with their duties, equipment and protocols so that when we finally did reach our first sampling station, we would be ready.
We arrived at our first station about 25 miles offshore where the depth was 25 meters (~75 feet), and everyone jumped into action as planned. I wasn’t expecting much at this station in terms of doliolids, but we were surprised to find what appeared to be a fairly large number of mature D. gengenbauri nurses. We caught them in our nets and saw them with the imager.

Live image of a D. gegenbauri nurse observed using the in situ mDPI imaging system at the DolMICROBE 25 m isobath station. (Inset) mDPI imager before deployment on the aft deck of the RV Savannah.
Over the next 5 hours we worked to collect samples and make measurements of the many parameters that will allow us to quantify the physical environment of the water column and to determine composition and activity of the microbial and zooplankton communities.

Sampling microbial parameters. Savannah Geiger filters water for DNA (top left), Laura Treible picking copepods for stable isotope analysis (top right), Kyle Aaron and Cristin Archer prepare the CTD for deployment to collect water (middle left), Anita Minniefield prepares filtration apparatus to collect RNA samples for transcriptomic analysis (middle right), Patrick Duffy and Laura Treible measure respiration (bottom left). Patrick Duffy incubating water samples for respiration measurements (bottom right).
Around dinner time we were finally ready to continue steaming out towards the outer edge of the shelf where our next sampling would occur. After another fabulous meal prepared by chef Ben (pot roast), we finally made it to our next station about 50 miles offshore where the depth was 45 meters (~ 150 feet). By this time, it was pretty late, so we decided to take a quick look at what was there and to start our measurements of respiration that require a long incubation, and call it a night.
Knowing it would take 10-11 hours to steam back to port, we woke-up early to sample the station. Especially because the weather continued to be kind to us, the sampling went as smoothly as it had the day before and a bit more quickly, because everyone was more familiar with their jobs. We finished sampling water quickly and finished processing the samples while we headed towards home. Because we didn’t find doliolids at this station and we did at the more inshore station, we stopped in between the two stations at the 35-meter isobath (~ 115 feet) to take a look there. Alas, we didn’t find doliolids there either, so we continued steaming home.
Because our next trip will also be the ship’s next trip, we were allowed to leave most of our gear on the ship so, after cleaning up a bit and putting away all the samples, we all headed home.
Over the next days, weeks, months and years, we will be processing and analyzing all the data we collected on this trip and the ones to come, slowly gathering enough data to answer some of the important ocean mysteries.
Our next trip is coming up quickly, so please stay tuned. In our future reports here, in addition to providing an overview of the dynamic ocean and what we encounter, we’ll focus this blog on describing the different measurements that we are making and why.
Signing off for now,
Marc




Thanks for sharing!! Great Content
Great Content!!
Really Helpful